By Karen Roman
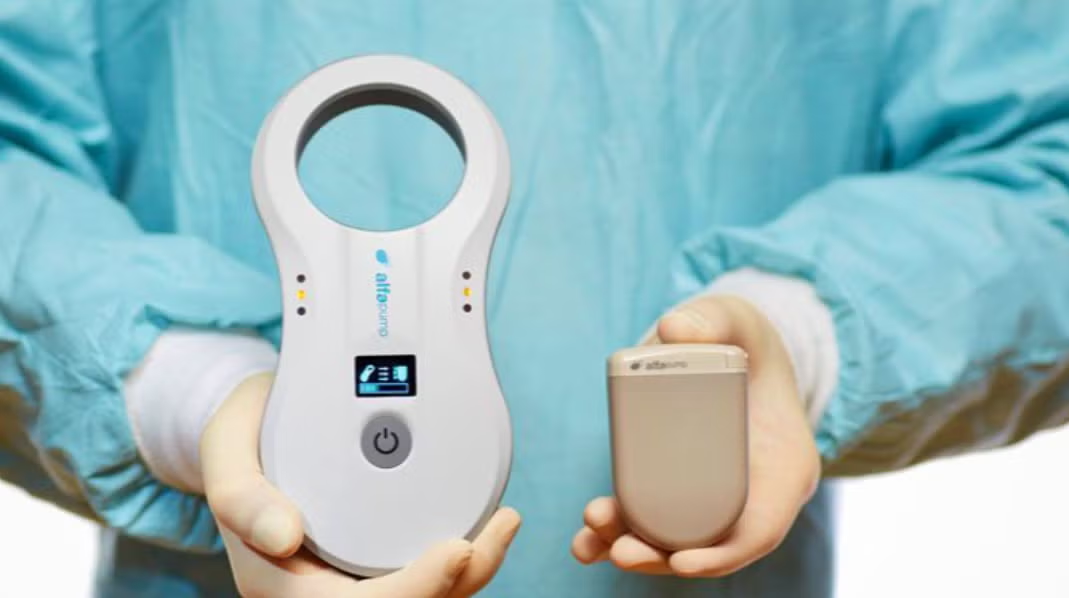
Sequana Medical said the company now has five medical centers using its alfapump, the first FDA-approved active implantable medical device to treat liver ascites due to cirrhosis.
The new implantations include the Dartmouth Hitchcock Medical Center and the University Medical Center of Southern Nevada, and both Mount Sinai Hospital and University of Pennsylvania have also implanted multiple patients, it stated.
“The medical centers that have already implanted multiple patients with the alfapump demonstrate the benefits they see to their patients and how it fits into their clinical practice as well as the overall value delivered to the healthcare system in reducing hospital visits and enabling patients to focus on living rather than managing their condition,” said Ian Crosbie, Sequana Medical CEO.
READ MORE
IPO Edge NYSE Firesides Scheduled for March 20 & June 2
Register for our weekly newsletter HERE
Contact:
Editor@IPO-Edge.com
Click HERE to follow us on LinkedIn






